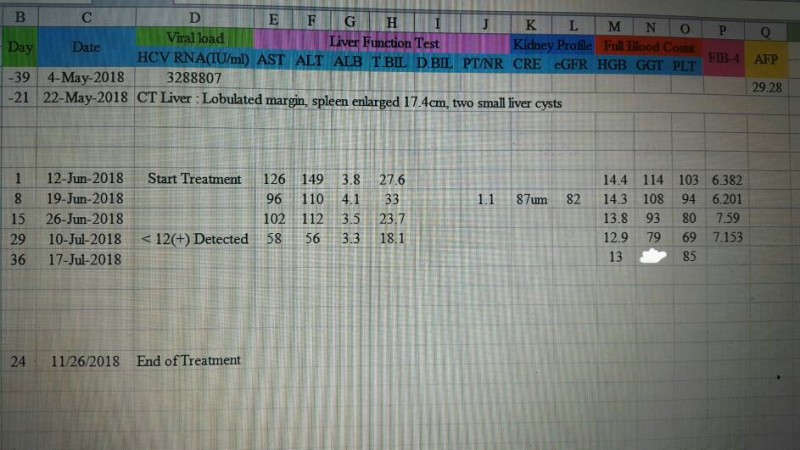
The main benefits of Ribavirin are at the start of treatment so my suggestion would be to use it while side effects can be tolerated (and haemoglobin and platelets remain ok).
If there are problems we can reduce the dose or stop it.
The extra treatment duration is much more valuable to success than ribavirin.
There is not a lot of good data about how successful treatment is in really sick patients (like Child Pugh
 www.hcvguidelines.org/unique-populations...ompensated-cirrhosis
www.hcvguidelines.org/unique-populations...ompensated-cirrhosis
The main guidance is from ASTRAL-4, where we saw:
www.nejm.org/doi/pdf/10.1056/NEJMoa1512614
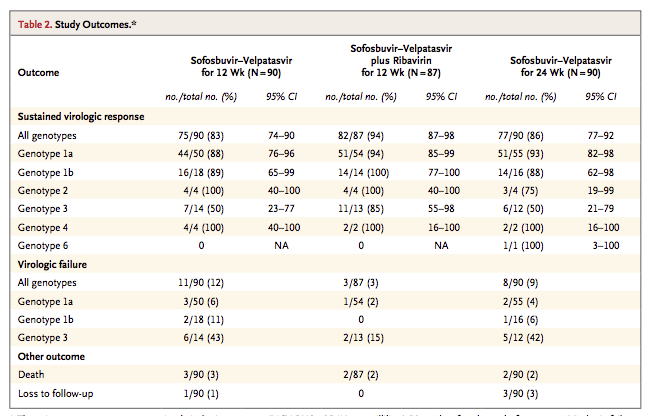
You will find this table in that paper from NEJM
As you can see from the GT3 patients the numbers are small so the confidence interval (margin for error) is quite wide.
For 12 weeks Sof/Vel without ribavirin success was 7/14 patients (50%)
With ribavirin that rose to 11/13 (85%) which is certainly better.
Unfortunately, Gilead did not do 24 weeks either with or without ribavirin but we know from experience what I have already told you - extra treatment duration provides a small, but very real, extra benefit.
www.clinicaloptions.com/hepatitis/confer.../capsules/206/page-1
I would estimate that with 24 weeks of treatment and at least 12 weeks with Ribavirin we can expect at least a 90% success rate - so 9 chances of success and only 1 chance of failure.
There is nothing better we could be giving. The NS3/4 boosters in Vosevii or Maviret (if combined with Sofosbuvir) or Zepatier (if combined with Sofosbuvir) can not be used in Child Pugh B or C.
You are, in short, doing everything that can possibly be done to maximise the chance of cure.