Epclusa is a prescription brand-name medication that’s used to treat hepatitis C virus (HCV) in adults. Epclusa contains two drugs: 100 mg of velpatasvir and 400 mg of sofosbuvir. It comes as a tablet you take by mouth once daily for 12 weeks.
Epclusa was approved in 2016 and was the first medication to treat all six hepatitis C genotypes. It can be used for people with or without cirrhosis (scarring of the liver). Epclusa is used to treat:
- people who have never been treated for HCV before (first-time treatment)
- people who have tried other HCV medications, but the drugs didn’t work for them
Studies show that Epclusa is effective for treating hepatitis C virus. In clinical trials, between 89 percent and 99 percent of people who received Epclusa achieved sustained virologic response (SVR). Achieving SVR means that the virus is no longer detectable in your body. The success rate varied depending on the person’s genotype (the strain of the virus they have) and medical history.
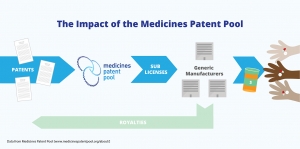
Epclusa contains the ingredients velpatasvir and sofosbuvir. There are generic forms available for Epclusa. Gilead released a generic Epclusa in early 2019 and Indian generics from companies such as Hetero, Mylan and Cipla have been available for some years
Epclusa can cause mild or serious side effects. The following list contains some of the key side effects that may occur while taking Epclusa. This list doesn’t include all possible side effects.
For more information on the possible side effects of Epclusa or tips on how to deal with a troubling side effect, talk with your doctor or pharmacist.
If you take Epclusa with ribavirin, you may have different or additional side effects. (See “Epclusa and ribavirin” below.)
More common side effects
The more common side effects of Epclusa can include:
- fatigue
- headache
- nausea
- insomnia (trouble sleeping)
- muscle weakness
- irritability
Less common side effects of Epclusa can include mild rash.
Most of these side effects may go away within a few days or a couple of weeks. If they’re more severe or don’t go away, talk to your doctor or pharmacist.
Serious side effects
Serious side effects from Epclusa aren’t common, but they can occur. Call your doctor right away if you have serious side effects. Call 911 if your symptoms feel life-threatening or if you think you’re having a medical emergency.
Serious side effects and their symptoms can include the following:
- Hepatitis B reactivation in coinfected patients. People who have both hepatitis C and hepatitis B can have a reactivation of the hepatitis B virus when they start taking Epclusa. This can occur even if the hepatitis B virus was treated in the past. Reactivation of hepatitis B can lead to liver failure and even death. Your doctor will test you for hepatitis B before you start treatment with Epclusa. If you have it, you may need to take medication to treat hepatitis B along with the Epclusa.
- Serious allergic reaction. Some people can have an allergic reaction after taking Epclusa. This is rare and usually not severe. However, some symptoms can be severe. Mild and severe symptoms of an allergic reaction can include:
- skin rash
- itchiness
- flushing (warmth and redness in your skin, typically in your face and neck)
- angioedema (swelling under your skin)
- swelling of your throat, mouth, and tongue
- trouble breathing
- Depression. Depression that occurred during clinical trials with Epclusa was mild to moderate and didn’t lead to any serious events. However, if you have symptoms of depression, be sure to call your doctor. Symptoms can include:
- feeling sad or hopeless
- trouble concentrating
- loss of interest in activities
Long-term side effects
Long-term side effects haven’t been reported with use of Epclusa. However, people with cirrhosis (liver scarring) may still experience symptoms of liver damage even after the hepatitis C virus is cleared from their body.
If you have cirrhosis, your doctor will want to monitor your liver function regularly, both during and after your treatment with Epclusa.
Side effects after treatment
Side effects after Epclusa treatment ends haven’t been reported in clinical studies.
There have been reports of people experiencing flu-like symptoms, such as fatigue, muscle aches, trouble sleeping, and chills, after treatment with Epclusa. However, these side effects are likely caused by your body recovering from the hepatitis C virus.
If you have flu-like symptoms after your Epclusa treatment ends, talk to your doctor.
Weight loss
Weight loss wasn’t reported as a side effect in a clinical study of Epclusa. However, some people experience weight loss as a symptom of hepatitis C. If you have weight loss that’s concerning or becomes severe, talk with your doctor.
Fatigue
Fatigue is a common side effect of Epclusa. In clinical studies, up to 22 percent of people who took Epclusa felt fatigue. This side effect may go away with continued use of the drug. If your fatigue is concerning or becomes severe, talk with your doctor.
Fatigue is also a side effect of hepatitis C. There are several ways to cope with hepatitis C fatigue. Staying hydrated, taking short naps, and eating healthy, well-balanced meals can help to fight tiredness. Regular exercise can also increase energy, so talk to your doctor about whether low-impact exercises are right for you.
Hair loss
Hair loss didn’t occur in clinical studies of Epclusa. Some people have reported losing hair during Epclusa treatment. However, it’s not clear if Epclusa was the cause of their hair loss.
Hair loss can be a symptom of hepatitis C. Your liver needs to be working well in order to get nutrients from the food you eat, and HCV prevents your liver from functioning properly. If you’re unable to obtain the nutrients your body needs, you may experience hair loss as a symptom of hepatitis C.
If you have hair loss and it becomes severe or concerning, talk with your doctor.
Depression
Depression is an uncommon side effect of Epclusa. In clinical studies, 1 percent of people who took Epclusa experienced mild to moderate depression.
Many people with hepatitis C can experience depression because of their diagnosis. If you feel depressed, talk with your doctor about ways to manage your mood.
As with all medications, the cost of Epclusa tablets can vary. To find current prices for Epclusa in your area, check out GoodRx.com:
The price you find on GoodRx.com is what you would pay without insurance. Your actual cost will depend on your insurance coverage.
Financial and insurance assistance
If you need financial support to pay for Epclusa or help understanding your insurance coverage, assistance is available.
Gilead Sciences Inc., the manufacturer of Epclusa, offers a program called Epclusa Support Path. For more information and to find out if you’re eligible for support, call 855-769-7284 or visit the program website.
Generic Epclusa
Drinking alcohol while taking Epclusa can increase your risk of certain side effects from the drug. These side effects include:
- headache
- nausea
- diarrhea
- fatigue
In addition, both hepatitis C and excessive alcohol use cause inflammation and scarring in the liver. The combination increases your risk of cirrhosis and liver failure.
Alcohol may also interfere with your ability to stick to your treatment plan as directed by your doctor. For instance, it may cause you to forget to take your medication on time. Missing doses could make Epclusa less effective in treating your HCV.
For all of these reasons, you should avoid drinking alcohol when you have hepatitis C, especially when you’re being treated with Epclusa. If you have trouble avoiding alcohol, talk with your doctor.
The following information describes the recommended dosage for Epclusa.
If you have decompensated cirrhosis (severe symptoms from advanced liver disease) or if you're genotype 3 with compensated cirrhosis or other medical conditions, you may also be prescribed ribavirin to take with Epclusa. The dosage of ribavirin you’re prescribed will depend on your weight, kidney function, and other health conditions. Your doctor can tell you more.
Drug forms and strengths
Epclusa is available in one strength. It comes as a combination tablet that contains 100 mg of velpatasvir and 400 mg of sofosbuvir.
Dosage for chronic hepatitis C
All people who take Epclusa to treat hepatitis C (HCV) take the same dosage. This dosage is one tablet taken once daily, preferably just before eating to aid with medication absorption.
How long will I take Epclusa?
You’ll take Epclusa once daily for 12 weeks.
What if I miss a dose?
It’s important to take Epclusa every day, to give you the best chance of curing your hepatitis C.
But if you do miss a dose, take it as soon as you remember. If it’s almost time for your next dose, only take one dose. Taking two doses at once can increase your risk of side effects.
Taking your Epclusa tablets exactly as your doctor prescribes is extremely important. That’s because following your treatment plan increases your chances of curing your hepatitis C (HCV). It also helps reduce your risk of long-term effects of HCV, such as cirrhosis and liver cancer.
Missing doses can interfere with how well Epclusa treats your hepatitis C. In some cases, if you miss doses, your HCV may not be cured.
So be sure to follow your doctor’s instructions and take one Epclusa tablet every day for 12 weeks. Using a reminder tool can be helpful in making sure you take Epclusa each day.
If you have any questions or concerns about your treatment, talk with your doctor. They can help resolve any issues for you and help you get the most out of your treatment.
The Food and Drug Administration (FDA) approves prescription drugs such as Epclusa to treat certain conditions.
Epclusa for chronic hepatitis C
Epclusa is FDA-approved for treating hepatitis C virus (HCV). It’s approved to treat all six main genotypes of HCV. Hepatitis C genotypes are different strains, or types, of the virus.
Epclusa is approved for use in people who have tried other HCV medications in the past and were unable to clear the virus. It’s also used for people who are new to HCV treatment.
People with or without cirrhosis are able to use Epclusa. Cirrhosis is serious scarring in the liver that prevents it from functioning properly. Epclusa is approved for use in people with compensated cirrhosis (liver disease that typically doesn’t cause symptoms) and decompensated cirrhosis.
Decompensated cirrhosis is when the liver is close to failure and causing serious health issues. People with decompensated cirrhosis will also need to take ribavirin (Rebetol) with Epclusa.
Other drugs are available that can treat hepatitis C. Some may be better suited for you than others. If you’re interested in finding an alternative to Epclusa, talk to your doctor to learn more about other medications that may work well for you.
Alternatives for treating hepatitis C
Epclusa is effective for treating hepatitis C caused by any of the six main genotypes. It can be used for people with or without cirrhosis, and for people with decompensated cirrhosis.
There are several other drugs and drug combinations that are used to treat hepatitis C. The drug regimen your doctor chooses for you will depend on your hepatitis C genotype and your liver function. It will also depend on whether or not you have received treatment for hepatitis C in the past.
Examples of other drugs that may be used to treat hepatitis C include:
- elbasvir and grazoprevir (Zepatier)
- glecaprevir and pibrentasvir (Mavyret)
- ledispavir and sofosbuvir (Harvoni)
- paritaprevir, ombitasvir, ritonavir, and dasabuvir (Viekira Pak)
- velpatasvir, sofosbuvir, and voxilaprevir (Vosevi)
- ribavirin (Rebetol), which is used in combination with other drugs
Interferons are medications that were commonly used to treat hepatitis C in the past. However, newer medications, including Epclusa, cause fewer side effects. Newer drugs also have higher success (cure) rates than interferons. Because of this, interferons are no longer typically used to treat hepatitis C.
You may wonder how Epclusa compares to other medications that are prescribed for similar uses. Here we look at how Epclusa and Harvoni are alike and different.
Epclusa contains two medications in one pill: velpatasvir and sofosbuvir. Harvoni also contains two drugs in one pill: ledipasvir and sofosbuvir.
Both medications contain the drug sofosbuvir, which is considered the “backbone” of the treatment. This means that the treatment plan is based on that drug, with other drugs added in combination.
Uses
Epclusa and Harvoni are both FDA-approved to treat hepatitis C. Epclusa is approved to treat all six genotypes of hepatitis C in adults with or without cirrhosis.
Harvoni is also approved to treat genotypes 1, 4, 5, and 6. It’s used for adults, and unlike Epclusa, it’s used for children aged 12 and older or who weigh at least 77 pounds.
This chart provides more detail on the hepatitis C genotypes that Harvoni is approved to treat:
| Coexisting conditions | Gen. 1 | Gen. 2 | Gen. 3 | Gen. 4 | Gen. 5 | Gen. 6 |
| Without cirrhosis | ✓ | ✓ | ✓ | ✓ | ||
| With compensated cirrhosis | ✓ | ✓ | ✓ | ✓ | ||
| With decompensated cirrhosis | ✓ | |||||
| Liver transplant recipient* | ✓ | ✓ |
* Epclusa can also be used in people who have received liver transplants, but it’s not approved by the FDA for that purpose.
Forms and administration
Epclusa and Harvoni are both taken as one tablet once daily. They are preferably taken just before eating to aid with medication absorption.
Epclusa is taken daily for 12 weeks. Harvoni is taken daily for either 12 or 24 weeks, depending on your liver function.
Side effects and risks
Epclusa and Harvoni belong to the same class of medications, so they have similar effects in the body. Therefore, they cause many of the same side effects. Below are some examples of these side effects.
More common side effects
More common side effects that can occur with both Epclusa and Harvoni include:
- fatigue
- headache
- nausea
- trouble sleeping
- muscle weakness
- irritability
In addition to these side effects, people taking Harvoni may also have:
- cough
- muscle pain
- shortness of breath
- dizziness
Serious side effects
Serious side effects that can occur with both Epclusa and Harvoni include:
- reactivation of the hepatitis B virus, which can lead to liver failure or death
- serious allergic reactions, including angioedema (severe swelling)
Boxed warnings
Epclusa and Harvoni both have boxed warnings from the FDA. A boxed warning is the strongest kind of warning that the FDA requires.
The boxed warnings describe a risk of reactivation of a hepatitis B infection after starting treatment with either drug. Reactivation of hepatitis B can lead to liver failure or death.
Your doctor will test you for hepatitis B before you start taking Epclusa or Harvoni. If the test results show that you have hepatitis B, you may need to take medication to treat it in order to prevent serious liver damage.
Effectiveness
Epclusa and Harvoni have been compared in clinical studies. Both are effective for treating hepatitis C, although Epclusa may cure a greater percentage of people than Harvoni does.
According to treatment guidelines, Epclusa and Harvoni are both first-choice medication options for the treatment of hepatitis C genotypes 1, 4, 5, and 6 in adults. In addition:
- Epclusa is a first-choice option for treating genotypes 2 and 3.
- Harvoni is a first-choice option for treating genotypes 1, 4, 5, and 6 in children ages 12 and older (or weighing 77 pounds and greater).
One that involved people with and without cirrhosis found that Epclusa and Harvoni had similar cure rates. It found that more than 93 percent of people who received ledipasvir and sofosbuvir, the components in Harvoni, were cured of the virus.
The cure rate for people taking velpatasvir and sofosbuvir, the components in Epclusa, was greater than 97 percent.
Another found that the two medications had similar rates of clearing the hepatitis C virus in people with compensated liver cirrhosis. And in a third , both drugs were again found to be highly effective at curing the virus.
However, in all three studies, Epclusa had slightly higher rates of SVR than Harvoni. SVR stands for sustained virologic response, which means undetectable virus levels after treatment.
Costs
Epclusa and Harvoni are brand-name medications. They are also available in generic forms. Generic drugs typically cost less than brand-name drugs.
The Gilead subsidiary Asegua Therapeutics released a generic Epclusa in early 2019. This has helped improve access. Buyers clubs such as FixHepC have been assisting patients to access affordable generic versions of Epclusa for some years.
Harvoni is typically more expensive than Epclusa. The actual price you pay for either drug will depend on your insurance plan.
Mavyret is another medication that’s used to treat hepatitis C. Here we look at how Epclusa and Mavyret are alike and different.
Epclusa contains two drugs in one pill: velpatasvir and sofosbuvir. Mavyret also contains two drugs in one pill: glecaprevir and pibrentasvir.
Uses
Epclusa and Mavyret are both FDA-approved to treat hepatitis C virus (HCV). They’re both used to treat all six genotypes in adults without cirrhosis or with compensated cirrhosis. However, Epclusa can also be used to treat people with decompensated cirrhosis, while Mavyret cannot.
Both medications can be used for people who are treating hepatitis C for the first time. They can also be used for people who have tried hepatitis C medications that didn’t work for them in the past.
Epclusa can be used for people who have tried any hepatitis C medication in the past. Mavyret, on the other hand, is only approved as a second treatment for people who have tried certain medications in the past. Your doctor can tell you if your past treatments make you eligible to take Mavyret.
Mavyret is also approved for use by people who have received liver or kidney transplants. Epclusa isn’t FDA-approved for use by people who have received these transplants, but in some cases, doctors may choose to prescribe the drug off-label for them.
Forms and administration
Epclusa and Mavyret both come as a single tablet that contains two drugs. You take one Epclusa tablet once daily preferably just before eating to aid with medication absorption. You take three Mavyret tablets together once daily. Mavyret should be taken with food.
Epclusa is taken for 12 weeks, while Mavyret is taken for 8, 12, or 16 weeks, depending on your medical history.
Side effects and risks
Epclusa and Mavyret have similar effects in the body and therefore cause very similar side effects. Below are examples of these side effects.
| Epclusa and Mavyret | Epclusa | Mavyret | |
| More common side effects | • headache • nausea • fatigue |
• trouble sleeping • weakness • irritability |
• diarrhea • itchy skin (in people on dialysis) • weakness (in people on dialysis) |
| Serious side effects | • serious allergic reactions • hepatitis B reactivation* |
(few unique serious side effects) | (few unique serious side effects) |
* Epclusa and Mavyret both have a boxed warning from the FDA for hepatitis B reactivation. A boxed warning is the strongest warning the FDA requires. It alerts doctors and patients about drug effects that may be dangerous.
Effectiveness
Epclusa and Mavyret haven’t been compared in clinical studies. However, both medications are highly effective at curing all genotypes of hepatitis C.
According to treatment guidelines, Epclusa and Mavyret are both first-choice options for the treatment of all six genotypes of hepatitis C. Your doctor may recommend one or the other based on your past treatment medications. The choice between the two drugs can also depend on your liver function.
In addition to these considerations, one of these drugs would be recommended over the other for certain medical conditions. These include:
- Severe chronic kidney disease: Mavyret is a first-choice option to treat hepatitis C in people with this condition. Epclusa, on the other hand, isn’t recommended for use in these people.
- Decompensated cirrhosis: For people with this condition, Epclusa can be used with ribavirin. However, Mavyret isn’t approved for people with decompensated cirrhosis.
Costs
Epclusa and Mavyret are brand-name medications. There are currently no generic forms of Mavyret. Brand-name medications usually cost more than generics.
The actual cost you pay for either drug will depend on your insurance plan.
Vosevi is another medication for hepatitis C that contains multiple drugs in one form. Epclusa contains the drugs velpatasvir and sofosbuvir in one tablet. Vosevi also contains velpatasvir and sofosbuvir in one tablet, but it also contains a third drug: voxilaprevir.
Uses
Epclusa and Vosevi are both FDA-approved to treat any of the six hepatitis C genotypes in adults without cirrhosis or with compensated cirrhosis. Epclusa is also approved to treat adults with decompensated cirrhosis.
Epclusa is approved to treat people who haven’t tried any hepatitis C regimen in the past, or who have tried treatments that didn’t work for them.
Vosevi, on the other hand, is approved to be used as a treatment for people who have tried only certain hepatitis C drugs and not had success with them. For instance, Vosevi is approved to treat:
- people with any genotype who have tried a certain type of antiviral called an NS5A inhibitor in the past
- people with genotypes 1a or 3 who have previously tried treatment that included sofosbuvir
If you’ve had hepatitis C treatment in the past, your doctor can tell you if this past medication use makes you eligible for treatment with Vosevi.
Forms and administration
Epclusa and Vosevi are both taken as one pill once daily. Epclusa should preferably be taken just before eating to aid with medication absorption, and Vosevi should be taken with food.
Both medications are taken for 12 weeks.
Side effects and risks
Epclusa and Vosevi are similar medications and cause many of the same side effects. Examples of these side effects are listed below.
More common side effects
More common side effects that can occur with both Epclusa and Vosevi include:
- headache
- fatigue
- nausea
- weakness
- trouble sleeping
In addition to these side effects, people taking Vosevi may also experience diarrhea.
Less common side effects that can occur with both Epclusa and Vosevi include mild rash.
Serious side effects
Serious side effects that can occur with both Epclusa and Vosevi include:
- reactivation of the hepatitis B virus*
- serious allergic reactions, including angioedema (severe swelling)
- depression
* Epclusa and Vosevi both have a boxed warning from the FDA for hepatitis B reactivation. A boxed warning is the strongest warning the FDA requires. It alerts doctors and patients about drug effects that may be dangerous.
Effectiveness
Epclusa and Vosevi have been directly compared in studies.
In one clinical study, Vosevi cured hepatitis C in more people than Epclusa. The researchers reported that 90 percent of people who took Epclusa for 12 weeks were cured of hepatitis C compared to 98 percent of people who took Vosevi.
Costs
Epclusa and Vosevi are both brand-name drugs. There are currently no generic forms of Vosevi, but buyers clubs such as FixHepC have been assisting patients to access affordable generic versions of Epclusa for some years.
Brand-name medications usually cost more than generics. Epclusa and Vosevi generally cost about the same. The actual cost you pay for either drug will depend on your insurance plan.
Epclusa can interact with several other medications. It can also interact with certain supplements.
Different interactions can cause different effects. For instance, some can interfere with how well a drug works, while others can cause increased side effects.
Epclusa and other medications
Below is a list of medications that can interact with Epclusa. This list doesn’t contain all drugs that may interact with Epclusa.
Before taking Epclusa, be sure to tell your doctor and pharmacist about all prescription, over-the-counter, and other drugs you take. Also tell them about any vitamins, herbs, and supplements you use. Sharing this information can help you avoid potential interactions.
If you have questions about drug interactions that may affect you, ask your doctor or pharmacist.
Amiodarone
Taking Epclusa with amiodarone (Pacerone, Nexterone) can cause bradycardia, which is a dangerously slow heart rate. This condition has also occurred with other medications that contain sofosbuvir, one of the components of Epclusa.
Some people who have taken amiodarone and sofosbuvir-containing medications have required a pacemaker in order to maintain a regular heart rate.
Taking amiodarone and Epclusa together isn’t recommended. If you have to take amiodarone while you receive Epclusa treatment, your doctor will closely monitor your heart function.
Digoxin
Taking Epclusa with digoxin (Lanoxin) can increase the amount of digoxin in your body. Increased digoxin levels can put you at risk of dangerous side effects.
If you need to take Epclusa and digoxin together, your doctor will closely monitor the amount of digoxin in your body. You may need a different digoxin dosage.
Certain cholesterol medications
Taking Epclusa with certain cholesterol medications called statins can increase the levels of statins in your body. This will put you at greater risk of side effects from these drugs, such as muscle pain and muscle damage.
Statins include drugs such as atorvastatin (Lipitor), rosuvastatin (Crestor), and simvastatin (Zocor). If you take Epclusa with a statin, your doctor will monitor you closely for signs of rhabdomyolysis (muscle breakdown).
To decrease your risk of side effects, you shouldn’t take Epclusa with a dose of rosuvastatin greater than 10 mg.
Certain seizure medications
Taking Epclusa with certain seizure medications can decrease the amount of Epclusa in your body. This may make Epclusa less effective. To avoid this interaction, don’t take Epclusa with these seizure medications.
Examples of seizure medications to avoid if you’re taking Epclusa include:
- carbamazepine (Carbatrol, Equetro, Tegretol)
- phenytoin (Dilantin, Phenytek)
- phenobarbital
- oxcarbazepine (Trileptal)
Topotecan
Taking Epclusa with topotecan (Hycamtin) can increase the level of topotecan in your body. This puts you at greater risk of topotecan’s side effects. Taking Epclusa with topotecan isn’t recommended.
Warfarin
Epclusa can affect your blood’s ability to form clots. If you take warfarin during your Epclusa treatment, your doctor may test your blood more often. Your warfarin dosage may need to be increased or decreased.
Certain HIV medications
Taking Epclusa with certain HIV medications can change your body’s levels of either Epclusa or the HIV drugs. These changes can make these drugs less effective or increase your risk of side effects.
Efavirenz
Taking Epclusa with efavirenz (Sustiva) can decrease levels of Epclusa in your body. This could make the medication less effective. To avoid this interaction, Epclusa and efavirenz shouldn’t be taken together.
Other medications that contain efavirenz should also be avoided. Examples of these drugs include:
- efavirenz, emtricitabine, and tenofovir (Atripla)
- efavirenz, lamivudine, and tenofovir (Symfi)
Tipranavir and ritonavir
Epclusa shouldn’t be taken with a combination of tipranavir (Aptivus) and ritonavir (Norvir). This combination of drugs will decrease Epclusa levels in your body. Lower Epclusa levels can make the medication less effective.
Tenofovir disoproxil fumarate
Taking Epclusa with HIV medications that contain tenofovir disoproxil fumarate will increase levels of tenofovir in your body. This increases your risk of tenofovir’s side effects, such as kidney damage.
If you take these medications with Epclusa, your doctor will closely monitor you for symptoms of tenofovir side effects. Examples of medications that contain tenofovir include:
- tenofovir (Viread)
- tenofovir and emtricitabine (Truvada)
- tenofovir, elvitegravir, cobicistat, and emtricitabine (Stribild)
- tenofovir, emtricitabine, and rilpivirine (Complera)
Epclusa and certain antibiotics
Certain antibiotic medications can decrease the amount of Epclusa in your body. Lower levels of Epclusa can make it less effective. To avoid this interaction, it’s recommended to avoid taking Epclusa with the following antibiotics:
- rifabutin (Mycobutin)
- rifampin (Rifadin, Rimactane)
- rifapentine (Priftin)
Epclusa and ibuprofen
There are no reported interactions between Epclusa and ibuprofen.
However, Epclusa shouldn’t be taken by people who have severe kidney disease. To avoid kidney damage caused by large doses of ibuprofen, don’t take a higher dose than is recommended on the ibuprofen package.
Epclusa and antacids
Taking Epclusa with antacids, such as Mylanta or Tums, can decrease the amount of Epclusa your body absorbs. This can cause low levels of Epclusa in your body, which may make Epclusa less effective.
To prevent this interaction, make sure twelve hours pass between taking antacids and your dose of Epclusa.
Epclusa and H2 blockers
Taking Epclusa with H2 receptor blockers can also decrease the amount of Epclusa that’s absorbed into your body. This may make Epclusa less effective.
To prevent this interaction, you should take Epclusa either at the same time as an H2blocker or 12 hours apart. Taking them at the same time allows both drugs to dissolve and be absorbed before the acid-reducing effects of the H2 blocker kicks in. Taking them 12 hours apart also allows each drug to be absorbed by your body without interacting with the other drug.
Examples of H2 blockers include famotidine (Pepcid), ranitidine (Zantac), and cimetidine (Tagamet HB).
Epclusa and PPIs
Taking Epclusa with proton pump inhibitors (PPI) can decrease the amount of Epclusa in your body. This could make Epclusa less effective. Taking Epclusa with a PPI isn’t recommended.
If you need to take a PPI while you are on Epclusa treatment, you should make sure twelve hours pass between taking antacids and your dose of Epclusa. Also, you should take Epclusa with food.
Examples of PPIs include:
- omeprazole (Prilosec)
- pantoprazole (Protonix)
- esomeprazole (Nexium)
- lansoprazole (Prevacid)
Epclusa and herbs and supplements
Taking Epclusa with St. John’s wort can decrease the amount of Epclusa that’s absorbed by your body. This may make Epclusa less effective. To avoid this interaction, don’t take Epclusa with St. John’s wort.
Other herbs or supplements that may decrease the amount of Epclusa in your body include:
Be sure to check with your doctor first before taking any new herbs or supplements during your treatment with Epclusa.
Epclusa is typically taken on its own to treat hepatitis C (HCV). However, in some cases it’s used with ribavirin (Rebetol).
Your doctor may prescribe ribavirin to be taken with Epclusa in the following situations:
- You have decompensated cirrhosis.
- You have a type of hepatitis C that’s resistant to medications (difficult to treat).
- You have failed treatment with other hepatitis C medications in the past.
- Your doctor prescribes it for off-label use (e.g., for treating HCV when you’ve had an organ transplant).
Epclusa and ribavirin are used together in people with these conditions because clinical studies showed a higher cure rate with the combination treatment.
Treatment with Epclusa and ribavirin lasts 12 weeks. Like Epclusa, ribavirin comes as a pill, but it’s taken twice a day. Typically, your dose of ribavirin will be based on your weight. It may also be based on your hemoglobin levels and your kidney function.
Ribavirin side effects and warnings
Ribavirin can cause several side effects and comes with some important warnings.
Boxed warning
Ribavirin has a boxed warning from the FDA. A boxed warning is the strongest kind of warning that the FDA requires. Ribavirin’s boxed warning advises that:
- Ribavirin shouldn’t be used alone to treat hepatitis C because it’s not effective by itself.
- Ribavirin can cause a type of blood toxicity called hemolytic anemia, which can lead to heart attack and death. For this reason, people who have serious or unstable heart disease shouldn’t take ribavirin.
- When used in pregnant women, ribavirin can cause serious harm or death to the fetus. Ribavirin shouldn’t be taken by pregnant women or their male sexual partner during pregnancy. Pregnancy should also be avoided for up to 6 months after the end of ribavirin treatment. During this time, consider using a backup method of birth control.
Other side effects
Ribavirin can also cause some common side effects, such as:
- fatigue
- headache
- nausea
- vomiting
- fever
- irritability
- muscle pain
Rare but serious side effects seen in clinical studies include anemia, pancreatitis, lung disease, and eye problems, such as infections and blurry vision.
Breastfeeding
It’s not known if ribavirin passes into human breast milk. Studies in animals showed that it may cause harm to nursing young. However, animal studies don’t always reflect how a drug would affect humans.
If you’re considering Epclusa treatment while breastfeeding, your doctor may recommend that you either stop breastfeeding or stop the treatment to avoid serious harmful effects.
You should take Epclusa according to your doctor’s instructions.
Timing
Epclusa can be taken at any time of day. If you experience fatigue during your treatment with the drug, taking it at night might help you avoid that side effect.
Taking Epclusa with food
Epclusa should preferably be taken just before eating to aid with medication absorption. Taking it with food might also help decrease any nausea that could be caused by the medication.
Can Epclusa be crushed?
It’s not known whether crushing Epclusa is safe. If you have trouble swallowing tablets, talk to your doctor about alternative medications rather than crushing your Epclusa tablets.
Epclusa is used to treat infection with the hepatitis C virus (HCV). Hepatitis C is a virus that’s transmitted through blood or body fluids. The virus primarily attacks cells in the liver. The resulting inflammation in the liver can lead to symptoms such as:
- pain in your abdomen (belly)
- fever
- dark urine
- joint pain
- jaundice (yellowing of your skin or the whites of your eyes)
Some people’s immune systems can create antibodies that fight the hepatitis C virus. However, many people will need to take medication to treat the virus and reduce long-term effects of the infection.
Serious, long-term effects of hepatitis C include cirrhosis (liver scarring) and liver cancer.
How Epclusa treats hepatitis C
Epclusa is a direct-acting antiviral (DAA) medication. DAAs treat HCV by preventing the virus from reproducing (making copies of itself). Viruses that can’t reproduce eventually die and are cleared from the body.
Clearing the virus from the body will reduce liver inflammation and prevent additional scarring.
How long does it take to work?
You may start to feel better days to weeks after you start taking Epclusa, but you’ll still need to take the full 12 weeks of treatment. It’s important to both take the full treatment and avoid missing any doses. These steps help the medication succeed in clearing the HCV from your body.
In clinical studies, more than 89 percent of people who took Epclusa were cleared of the virus after three months of treatment. Your doctor will test you before and during treatment with Epclusa, and again 12 weeks after you finish taking Epclusa. This last test will determine if you are “cured” of hepatitis C.
You’re considered cured of hepatitis C when you have achieved sustained virologic response (SVR), which means the virus is no longer detectable in your blood.
There haven’t been enough studies in humans to know if Epclusa is safe to take during pregnancy. Studies in animals didn’t show harm to the fetus when the mother received the drug. However, animal studies don’t always predict what would happen in humans.
If you become pregnant while taking this drug, call your doctor right away.
Note: If you’re taking Epclusa with ribavirin, that combination treatment can be dangerous to a pregnancy (see “Epclusa and ribavirin” above).
It isn’t known if Epclusa passes into breast milk in humans. In animal studies, Epclusa was found in breast milk, but it didn’t cause harmful effects. However, animal studies don’t always reflect what will happen in humans.
If you’re breastfeeding your child and considering taking Epclusa, talk with your doctor about the potential risks and benefits.
Note: If you’re taking Epclusa with ribavirin, you should talk to your doctor about whether you can safely continue breastfeeding (see “Epclusa and ribavirin” above).
Here are answers to some frequently asked questions about Epclusa.
Does stopping Epclusa cause withdrawal symptoms?
Epclusa hasn’t caused withdrawal symptoms in people in clinical studies.
There have been reports of people having flu-like symptoms after treatment with Epclusa. These symptoms include fatigue, muscle aches, and headache. However, it’s more likely that these side effects would be caused by your recovery from the hepatitis C virus.
If you have flu-like symptoms after you finish your Epclusa treatment, talk to your doctor.
How long will Epclusa take to get rid of my hepatitis C?
Epclusa will start working right away, but it will take several weeks to a few months to achieve a sustained virologic response (SVR). Achieving SVR means that the virus is no longer detectable in your body.
You’ll take Epclusa for 12 weeks, and 12 weeks after you finish treatment, your doctor will test your blood. At this time, SVR is achieved in most cases. Essentially, this means that your HCV infection has been cured.
What is the cure rate for Epclusa?
In clinical studies, between 89 percent and 99 percent of people who received Epclusa were cured of the virus. Cure rates were slightly different based on genotype, liver function, and previous hepatitis C treatment.
Can hepatitis C come back after taking Epclusa?
If you take Epclusa for the 12 weeks of treatment as your doctor prescribed and reach SVR 24 (undetectable virus levels 24 weeks after finishing treatment), then maintain a healthy lifestyle and avoid routes of reinfection, the virus won’t come back.
It’s possible to become reinfected with the virus after taking any hepatitis C medication, including Epclusa. Reinfection can happen in the same way the original infection was contracted. Sharing needles used for injecting drugs and having sex without using a condom are possible routes of reinfection.
However, avoiding these behaviors can help prevent reinfection with hepatitis C.
What is a hepatitis C genotype?
There are six different types, or strains, of hepatitis C viruses that infect people. These strains are called genotypes.
Genotypes are identified by differences in the genetic code of the viruses. Genotype 1 is the most common hepatitis C strain in the United States, but other strains are also seen in this country.
Your doctor will do a blood test to figure out which genotype you have. Your hepatitis C genotype will help your doctor choose the right medication for you.
Can I take Epclusa if I have HIV as well as hepatitis C?
Yes, you can. Epclusa can be safely used to treat hepatitis C in people who are also infected with HIV.
In a clinical study involving people who had both hepatitis C and HIV, up to 95 percent of people who received Epclusa were cured of hepatitis C. Importantly, treatment with Epclusa didn’t cause worsening of HIV.
FDA warning: Reactivation of HBV infection
- This drug has a boxed warning. This is the most serious warning from the Food and Drug Administration (FDA). A boxed warning alerts doctors and patients about drug effects that may be dangerous.
- Reactivation of hepatitis B virus (HBV) can occur in people who are coinfected with both hepatitis C virus and HBV. This can happen during or after treatment with Epclusa. Your doctor will do blood tests for hepatitis B before you start taking Epclusa. If you currently have hepatitis B or have had it in the past, you may need to take medication for HBV.
Other warnings
Before taking Epclusa, be sure to tell your doctor about your health history. This is especially important if you have severe kidney disease.
It isn’t known if Epclusa is safe or effective in people with severe kidney disease. This includes people with severely reduced kidney function, or with end-stage kidney disease that requires hemodialysis.
If you have severe kidney disease, Epclusa may not be the right medication for you.
Taking too much Epclusa can increase your risk of serious side effects.
Overdose symptoms
Symptoms of an overdose can include:
- severe nausea
- headache
- muscle weakness
- fatigue
- trouble sleeping
- irritability
What to do in case of overdose
If you think you’ve taken too much of this drug, call your doctor or seek guidance from the American Association of Poison Control Centers at 800-222-1222 or through their online tool. But if your symptoms are severe, call 911 or go to the nearest emergency room right away.
When Epclusa is dispensed from the pharmacy, the pharmacist will add an expiration date to the label on the bottle. This date is typically one year from the date the medication was dispensed.
The purpose of such expiration dates is to guarantee the effectiveness of the medication during this time.
The current stance of the Food and Drug Administration (FDA) is to avoid using expired medications. However, an FDA showed that many medications may still be good beyond the expiration date listed on the bottle.
How long a medication remains good can depend on many factors, including how and where the medication is stored. Epclusa pills should be stored at temperatures up to 86°F (30°C) and kept in the original container.
If you have unused medication that has gone past the expiration date, talk to your pharmacist about whether you might still be able to use it.
The following information is provided for clinicians and other healthcare professionals.
Mechanism of action
Epclusa contains two drugs: velpatasvir and sofosbuvir.
Velpatasvir inhibits HCV NS5A protein, which is hypothesized to be necessary for efficient phosphorylation of viral RNA. Inhibition of NS5A blocks RNA replication and assembly.
Sofosbuvir is an HCV NS5B polymerase inhibitor with an active metabolite (a nucleoside analog triphosphate) that is incorporated into HCV RNA. The active metabolite acts as a chain terminator, halting HCV replication.
Epclusa has activity against all six main HCV genotypes.
Pharmacokinetics and metabolism
Epclusa contains two active ingredients: velpatasvir and sofosbuvir.
Velpatasvir reaches peak concentration in about three hours and is almost completely bound to plasma proteins. It is metabolized by CYP2B6, CYP2C8, and CYP3A4 enzymes. The half-life is about 15 hours, and it is eliminated primarily in the feces.
Sofosbuvir’s peak concentration occurs in 30 minutes to one hour. Plasma protein binding accounts for approximately 65 percent of the circulating drug.
Sofosbuvir is a prodrug that is converted to an active metabolite (GS-461203) by hydrolysis and phosphorylation in the liver. GS-461203 is further dephosphorylated to an inactive metabolite. Up to 80 percent of the dose is eliminated in the urine. The parent drug’s half-life is 30 minutes, and the metabolite’s half-life is around 25 hours.
Both components of Epclusa are substrates of P-gp and BCRP.
Contraindications
There are no contraindications to Epclusa use. Refer to ribavirin contraindications for patients receiving the Epclusa and ribavirin combination regimen.
Storage
Epclusa should remain in the original packaging. The container should be stored below 86F (30C).
Disclaimer: Healthline has made every effort to make certain that all information is factually correct, comprehensive, and up-to-date. However, this article should not be used as a substitute for the knowledge and expertise of a licensed healthcare professional. You should always consult your doctor or other healthcare professional before taking any medication. The drug information contained herein is subject to change and is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. The absence of warnings or other information for a given drug does not indicate that the drug or drug combination is safe, effective, or appropriate for all patients or all specific uses.