This is a screenshot of an article in the Australian today. Here is a link to the full text of the study published in the British Medical Journal http://bmjopen.bmj.com/content/5/11/e009758.full.pdf And topping the list is..... No prizes for guessing.
So Big Pharma's argument about pricing goes like this: We spend a lot on R&D so we need to make a lot back when we find a winner. Now with Pharmasset we can look at the balance sheet immediately prior to Gilead's…
You've almost certainly seen a version of this picture. It's called earthrise and really hammers home the idea that we live co-operatively on a tiny speck in a dark black void. This particular version was taken out of the window of Apollo…
Rumour has it that PBS listing of HCV DAAs is not going to happen this year. It's not official yet, but... We've started treatment for 5 patients on the transplant list in the last week, all referred from various centers. If you're…
Thursday, 22 October 2015 00:05
It is not just business, it's personal - I have failed, but I will do better
Written by Super User
I just sent an email called "There are no words" because... there simply are not. Dear L, Please accept my condolences about M's passing and know that the opportunity to meet such a beautiful human being, who became so suddenly unwell, and…
We have previously tested Incepta's Twinvir and today have received samples of Beacon Pharma's Sofosbuvir, Daclatasvir and Ribavirin products for testing. We will share the testing results as soon as they are available. The quality of production of the packaging is extremely…
Here is some interesting data from the Google Analytics about the FixHepC visitor demographics. It's kind of spooky that Google knows your age and sex, but here's their analysis. One of the interesting things is that with 1% of the population infected…
Tagged under
NSW Health and ASHM (Australian Society of HIV/HBV/HCV Medicine) have joined forces to issue a position statement titled Importation of Generic HCV Drugs. You can follow the link or read the text below: 08 October 2015 Advice for HCV Clinicians This communique…
Tagged under
Lismore Liver Clinic is now able to accept referrals for assessment and monitoring of patients accessing generic medications for the treatment of hep C. Please note that this does not represent an endorsement of the use of generics, but rather an acceptance…
Tagged under
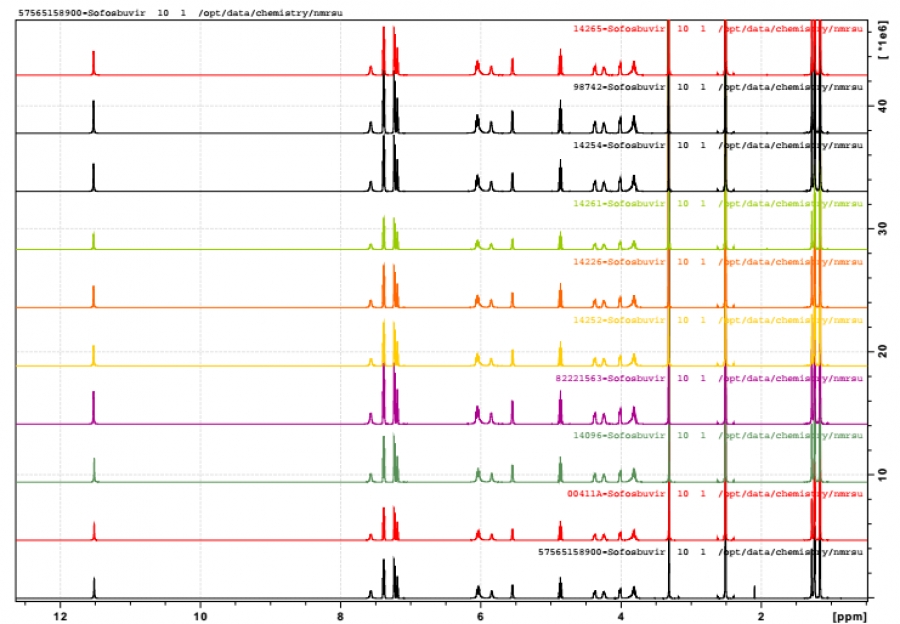
So what is this image? It is a patient safety check. Each coloured line represents a single patient's self imported Sofosbuvir being tested against a reference standard. Yes it's just lines but for each chemical there is a unique NMR signature represented…
Tagged under
Tuesday, 06 October 2015 19:28
The Trans-Pacific Partnership: the dirtiest trade deal, you've never heard of
Written by Super User
So last night the TPP got the nod in the US. So what is it and what does it mean? It's supposed to be a free trade agreement, but it apparently has some catches, particularly around medications and patents. Sadly it's actually…
The thing with Hep C is that it's not about anonymous statistics, it's about real people. Here's the story of one patient, and his wife's journey through Interferon and failure and then to self initiated treatment and cure via the parallel import…
Tagged under
Saturday, 03 October 2015 20:12
The Minimum Cost To Cure Hepatitis C - Revisited
Written by Super User
A patient posted a link that contained a powerpoint presentation from Dr Andrew Hill, PhD. I asked Dr Hill if I could post it here and he said yes. I turned that into a very quick YouTube movie, so you will probably…
Tagged under
A story about the genesis of this initiative titled Big pharmaceuticals and the Hepatitis C drug trail dubbed a 'miracle cure' ran this morning in The Sydney Morning Herald and The Age. http://www.smh.com.au/nsw/big-pharmaceuticals-and-the-hepatitis-c-drug-trail-dubbed-a-miracle-cure-20150924-gjtupf.html There is also another story in the Sydney Morning…
Tagged under
Hepatitis Australia is using new data released from the Kirby Institute today showing only one per cent of people with hep C received treatment last year to push the government to list new hep C drugs without delay. "It's time for action,"…
Tagged under
It has always been a worry that the powers that be may not like the idea of patients parallel importing life saving medications. The Buyers Club has been active for less than 24 hours and already it seems the battle lines are…
Tagged under
In the last blog reference was made to the failed 2014 PBAC submission, simply because I could not find the PBAC outcome. Somebody has been kind enough to flag my attention to the PBAC 2015 recommendation papers which you can read here:…
In Australia there has been optimism about the PBACs decision to recommend that Hep C treatments are put on the PBS. Some people seem to think that what this means is that these new medications may well be widely available at affordable…
So I received an email pointing out that you can make these new medications for $1 a dose. The question, not really a question, more of a talking point, was this: Hill et al 2014: US$2000 might seem dirt cheap for a…
Assisted by the Federal Investigation Authority (FIA), personnel of the Drug Regulatory Authority of Pakistan (DRAP) raided one of the local industries in Islamabad’s Kahuta industrial area here Saturday, leading to the discovery of unlawful manufacturing of Sofasbuvir tablets, which are used…
Friday, 08 May 2015 19:31
WHO moves to improve access to lifesaving medicines for hepatitis C
Written by Super User
GENEVA - WHO today published the new edition of its Model List of Essential Medicines which includes ground-breaking new treatments for hepatitis C. http://www.who.int/mediacentre/news/releases/2015/new-essential-medicines-list/en/
Tagged under
Monday, 01 June 2015 13:49
Drug Safety Update: Risks of severe bradycardia and heart block when taken with amiodarone
Written by Super User
On the 20th May 2015 the Medicines and Healthcare products Regulatory Agency in the UK issued a Drug Safety Update titled: Sofosbuvir with daclatasvir; sofosbuvir and ledipasvir: risks of severe bradycardia and heart block when taken with amiodarone. You will find a…
Tagged under
Today we compared a new batch of Sofosbuvir and Ledipasvir to our reference sample data and found it consistent. It's reassuring to see the consistency.
Tagged under
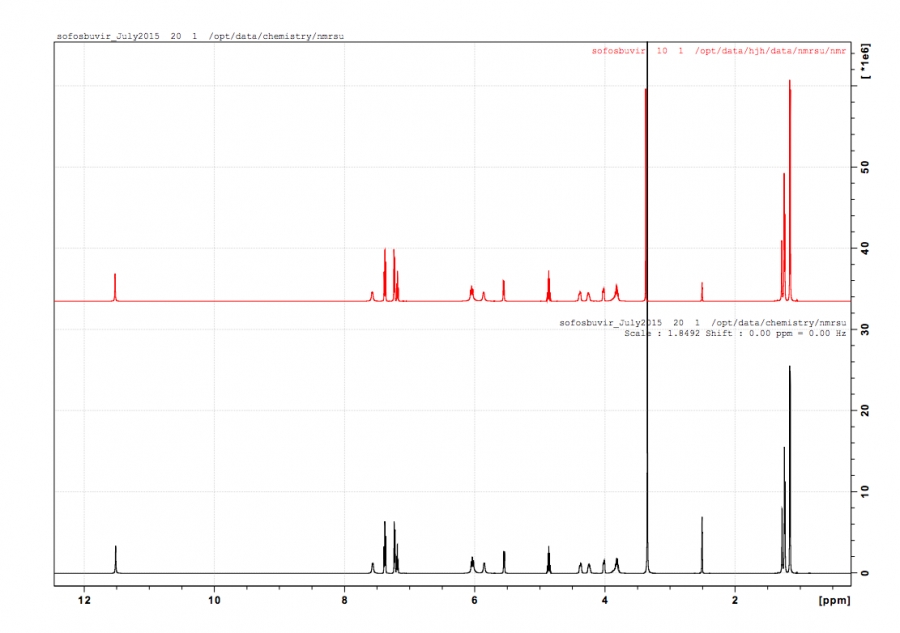
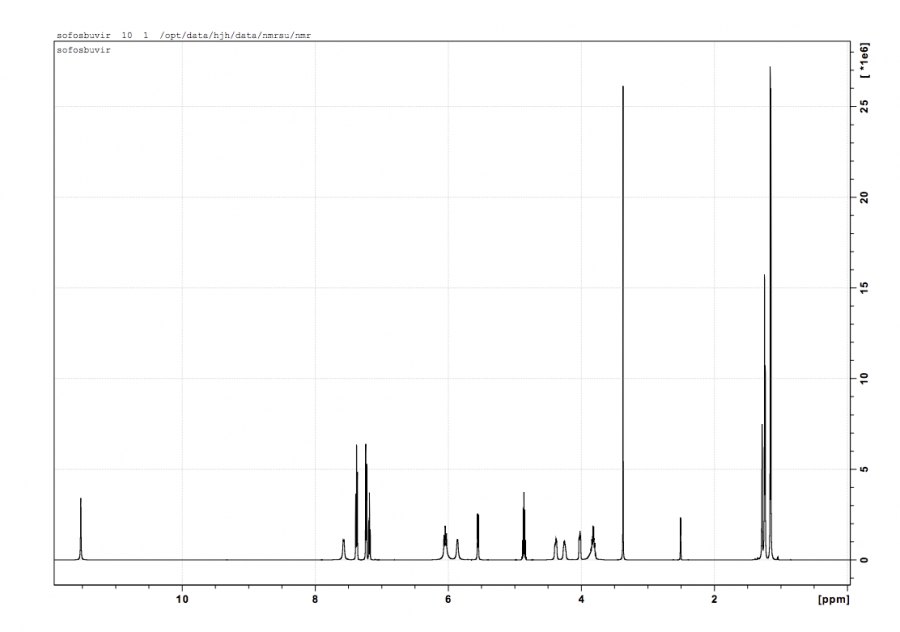
We were quite excited to see the first shipment of Chinese sourced Sofosbuvir and Ledipasvir. A white powder and and off white powder, but what would the NMR show? Here's the NMR for Sofosbuvir which is right on the money. Ledipasvir: mass…
Tagged under
More...
In Australia the PBAC has voted to recommend the inclusion of sofosbuvir in the PBS. This could mean that this vital medication becomes available at affordable prices in Australia, however it's also possible Cabinet will reject this given that inclusion in the…
Tagged under